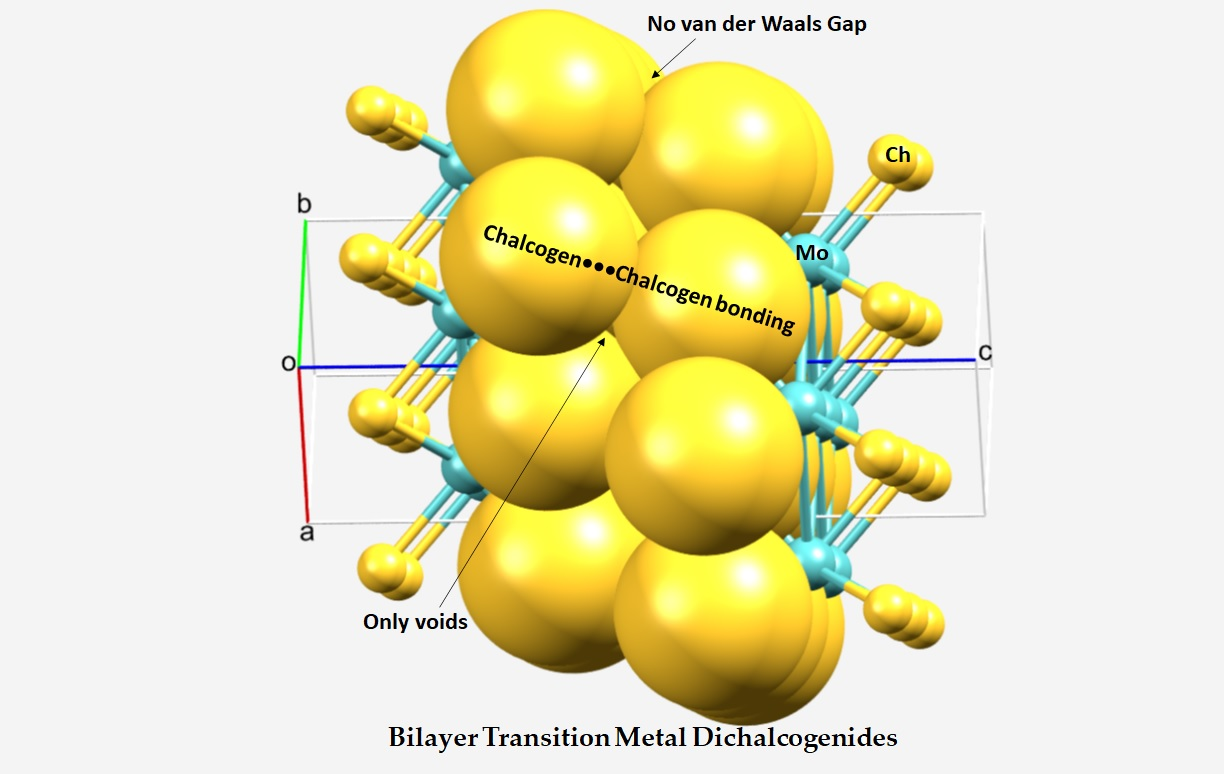
Figure 2 from Structure and bonding of Au5M (M=Na, Mg, Al, Si, P, and S) clusters | Semantic Scholar
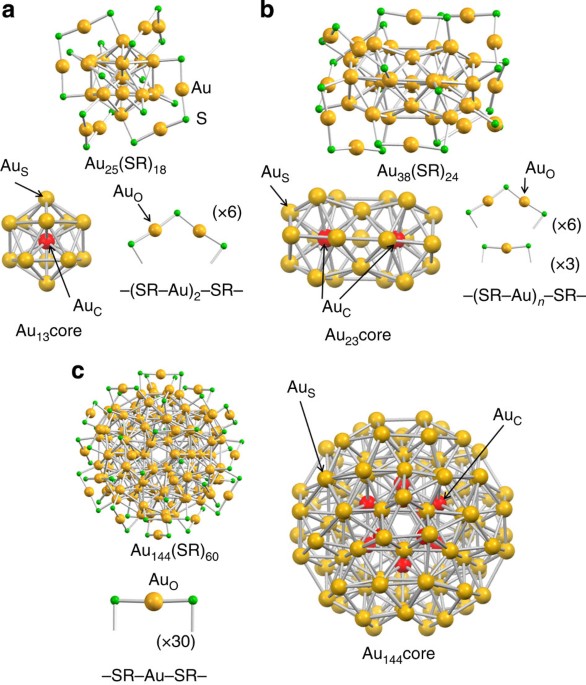
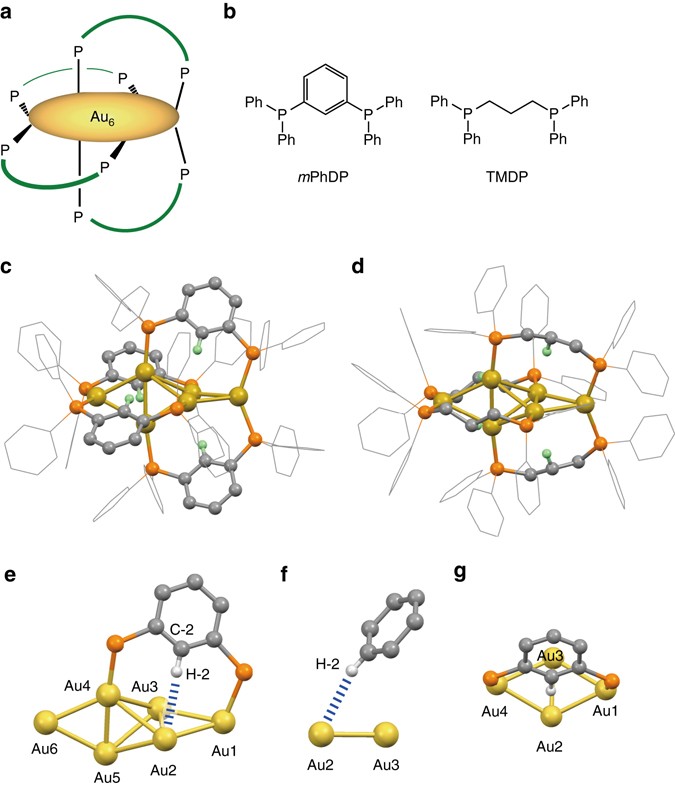
Hierarchy of bond stiffnesses within icosahedral-based gold clusters protected by thiolates | Nature Communications
1:50 explain how the structures of diamond, graphite and C60 fullerene influence their physical properties, including electrical conductivity and hardness - TutorMyself Chemistry

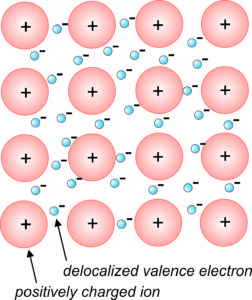
Metallic Bonds Quartz. Metal atoms are arranged in very compact and orderly patterns. Although metals do not bond ionically, they often form lattices. - ppt download