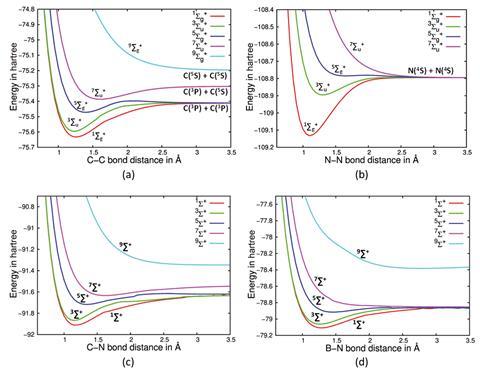
Excited state potential energy curves reignite diatomic carbon's bond order conundrum | Research | Chemistry World
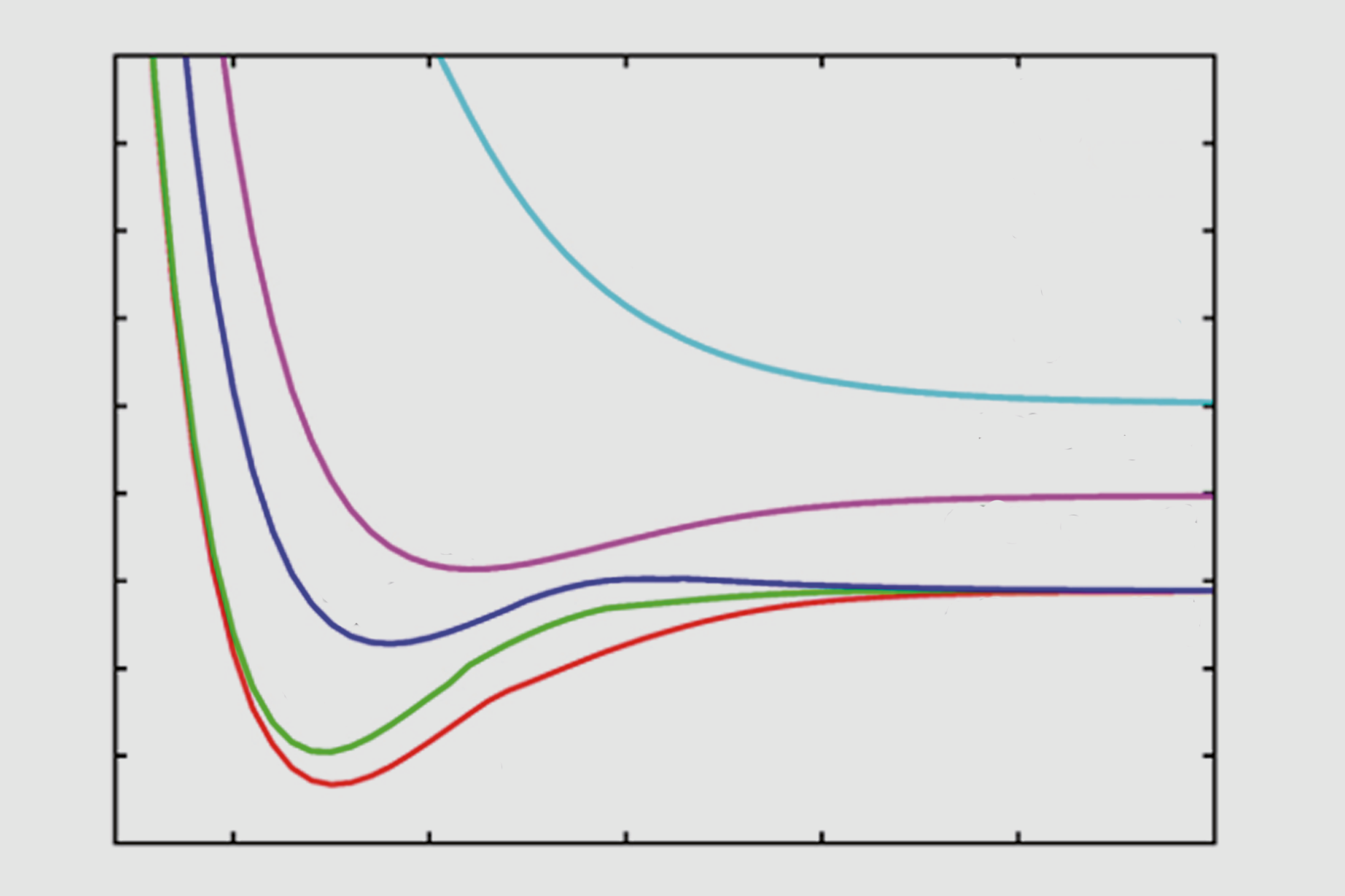
Excited state potential energy curves reignite diatomic carbon's bond order conundrum | Research | Chemistry World

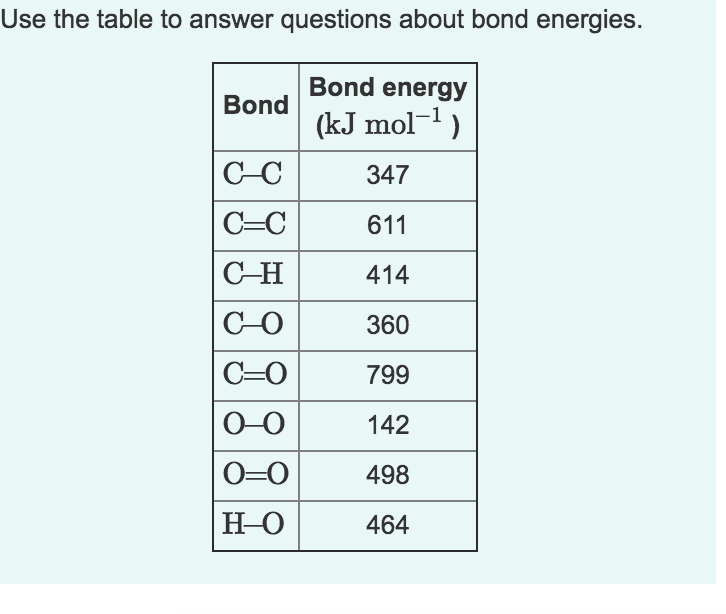
12 Methane burns in oxygen to form carbon dioxide and water. CH4(9) + 2O2(g) → CO2(g) + 2H2O(1) The bond energies are shown in the table. bond bond energy in kJ/mol 410
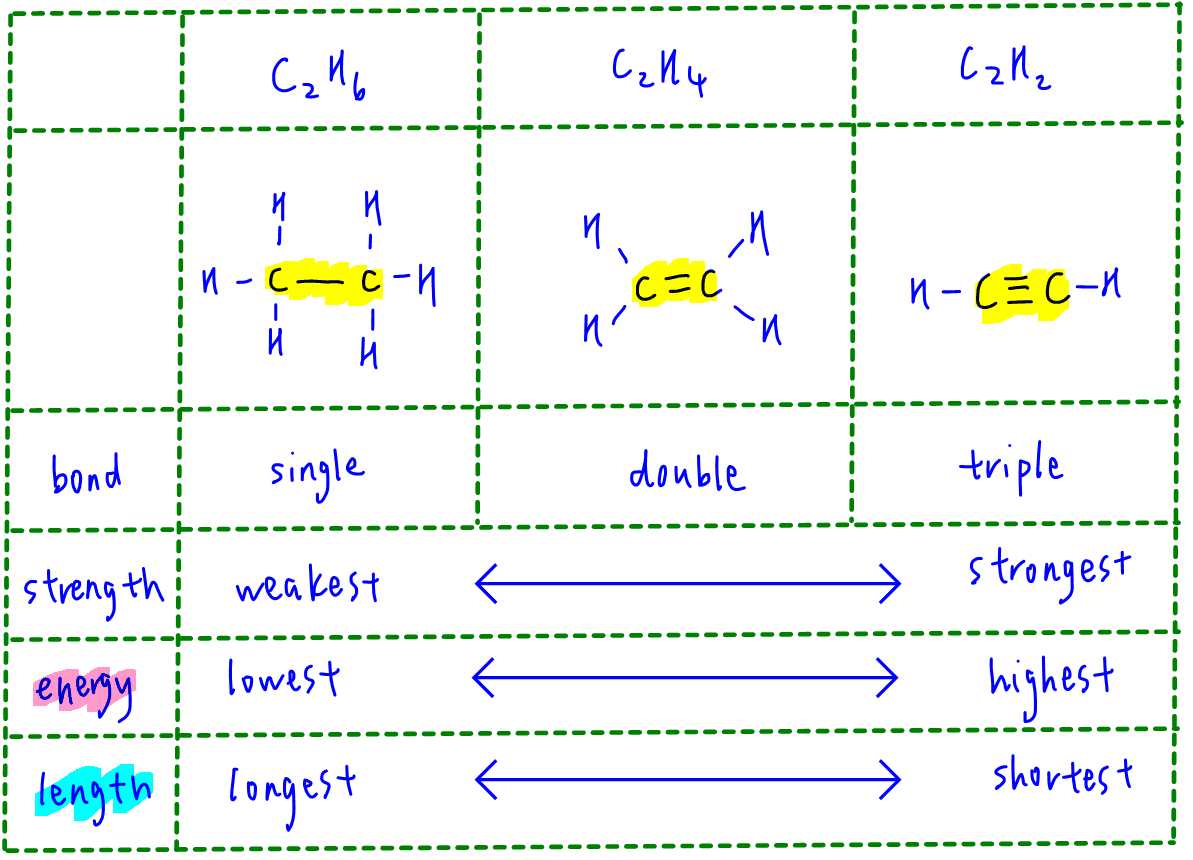
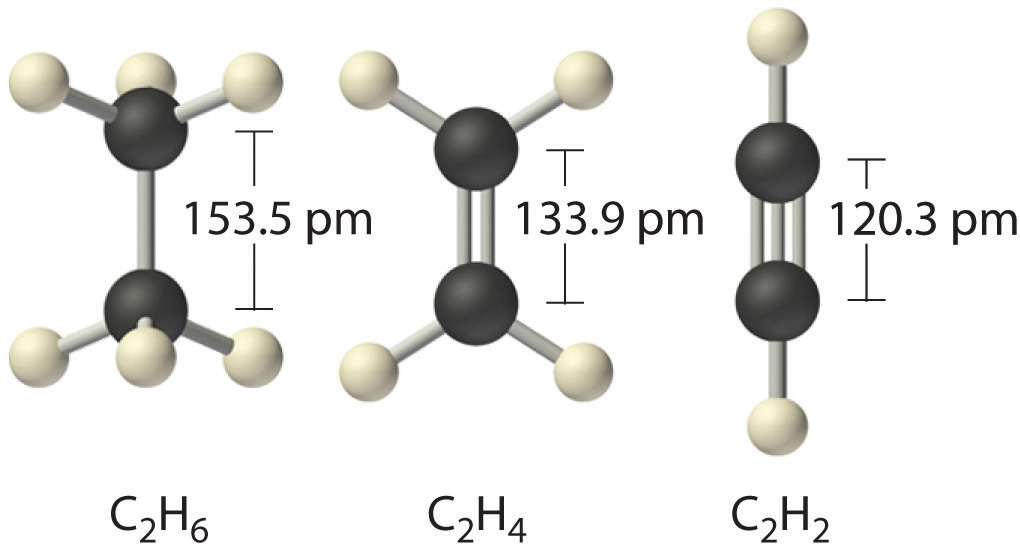
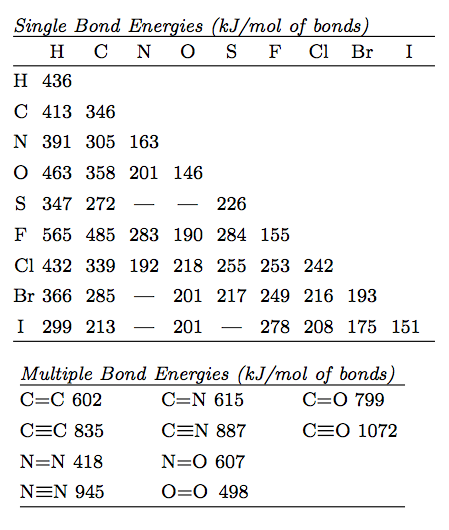
Which of the following has the strongest carbon-carbon bond ?C_{2}H_{2}C_{2}H_{4}C_{2}H_{6}C_{3}H_{8}C_{3}H_{6}






.jpg?revision=1)