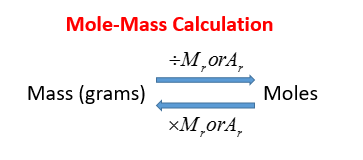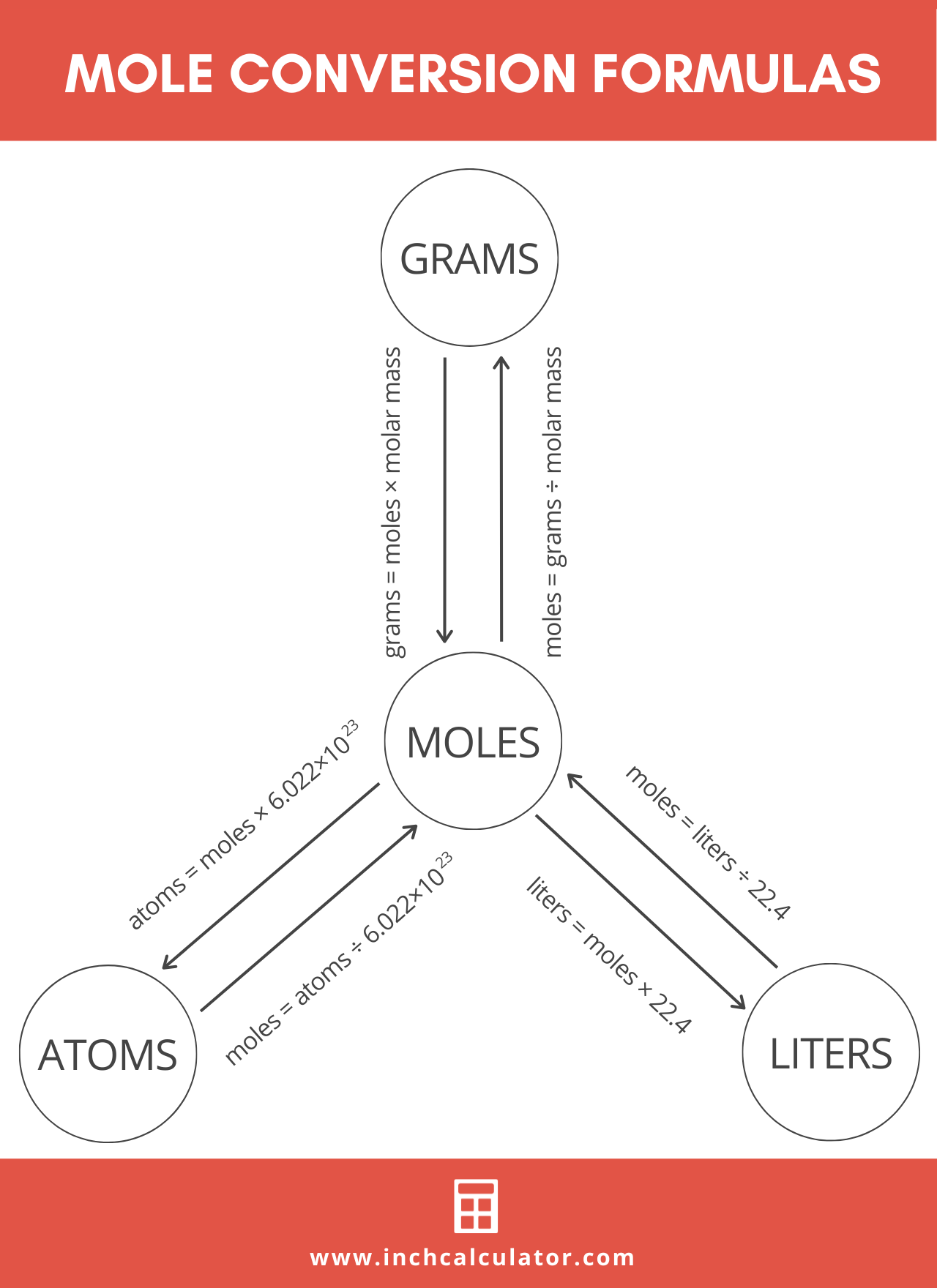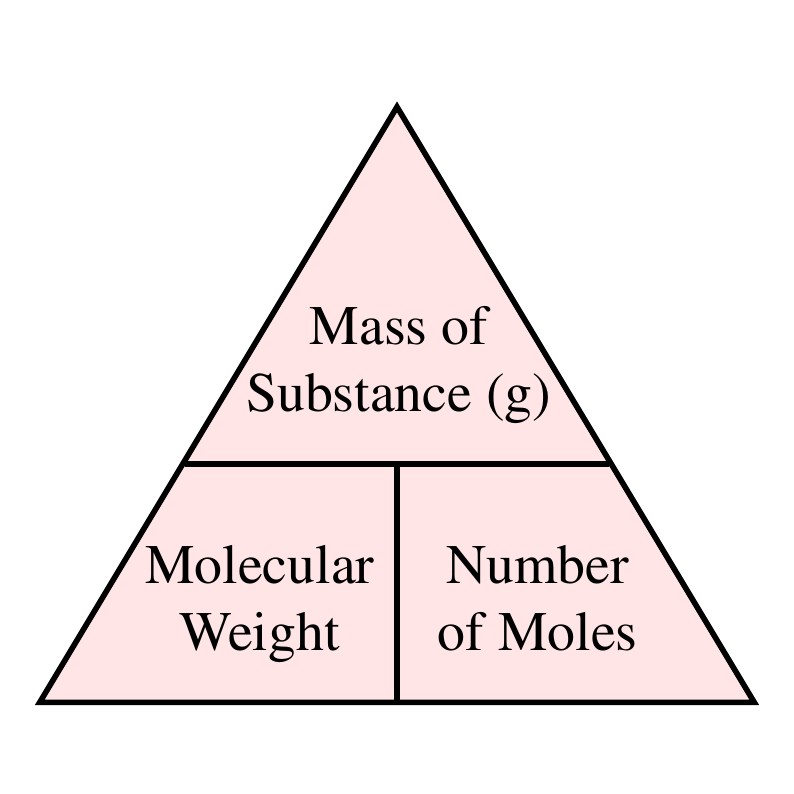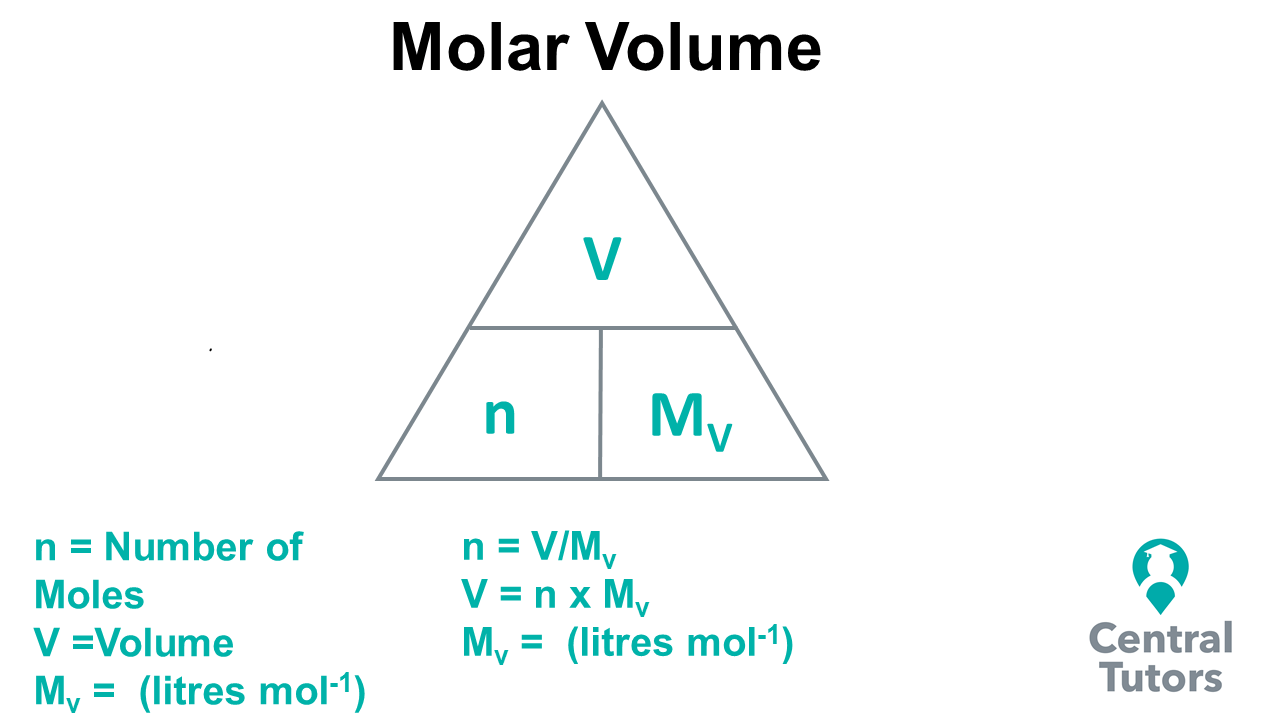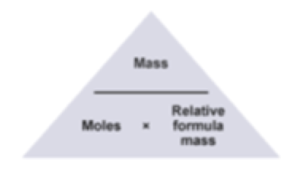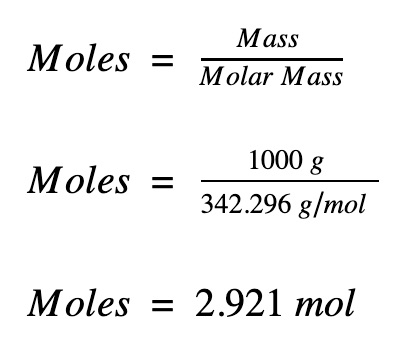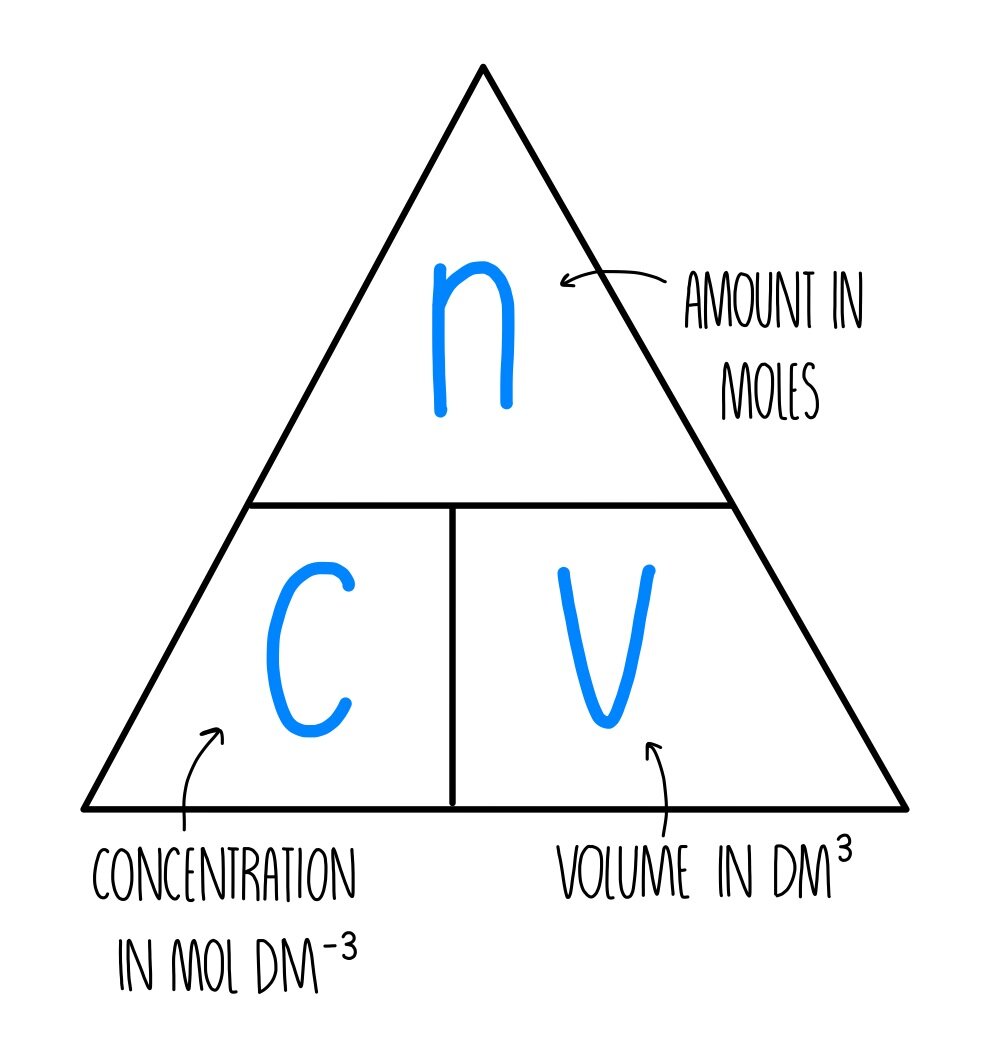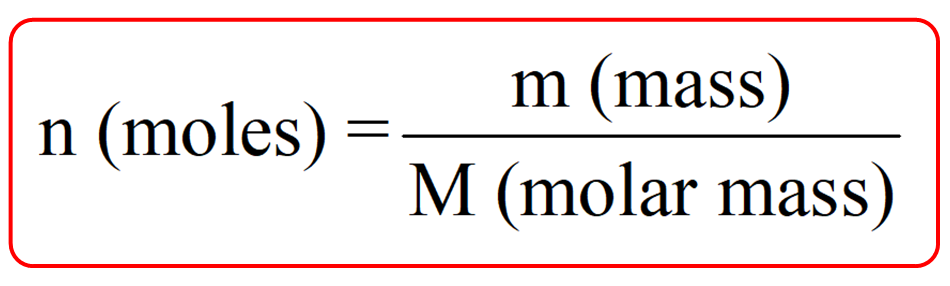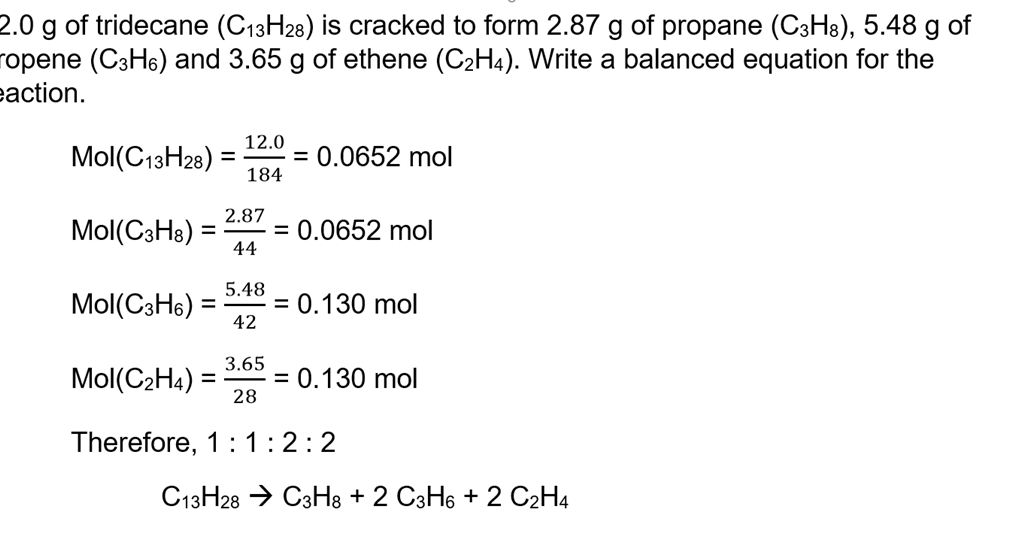
The mole formula triangle or pyramid isolated on a white background. Relationship between moles, mass, and molar mass with equations. Mass-mole calculation – n=m/Mr. Triangle used in chemistry. Stock Vector | Adobe
Mole calculations in solutions - Higher - Titration and calculations - GCSE Chemistry (Single Science) Revision - WJEC - BBC Bitesize

molar gas volume Avogadro's Law moles and mass calculations gcse chemistry calculations igcse KS4 science A level GCE AS A2 O Level practice questions exercises

definition mole explained molar mass mol mols calculations how to read equations in moles deducing equations from reacting mole ratios questions gcse chemistry igcse KS4 science A level GCE AS A2 O
Mole calculations - The mole and concentration of solutions - National 5 Chemistry Revision - BBC Bitesize