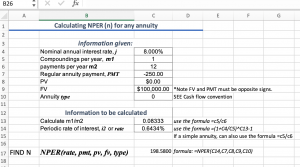
Calculate the number of particles in each of the following: (a) 48 g of Mg (b) 8 g of O2 (c) 0.1 mole of carbon (Atomic mass Mg = 24 u, O =

Calculate the number of protons, electrons, neutrons in Bromine with atomic number 35 and mass number 80.

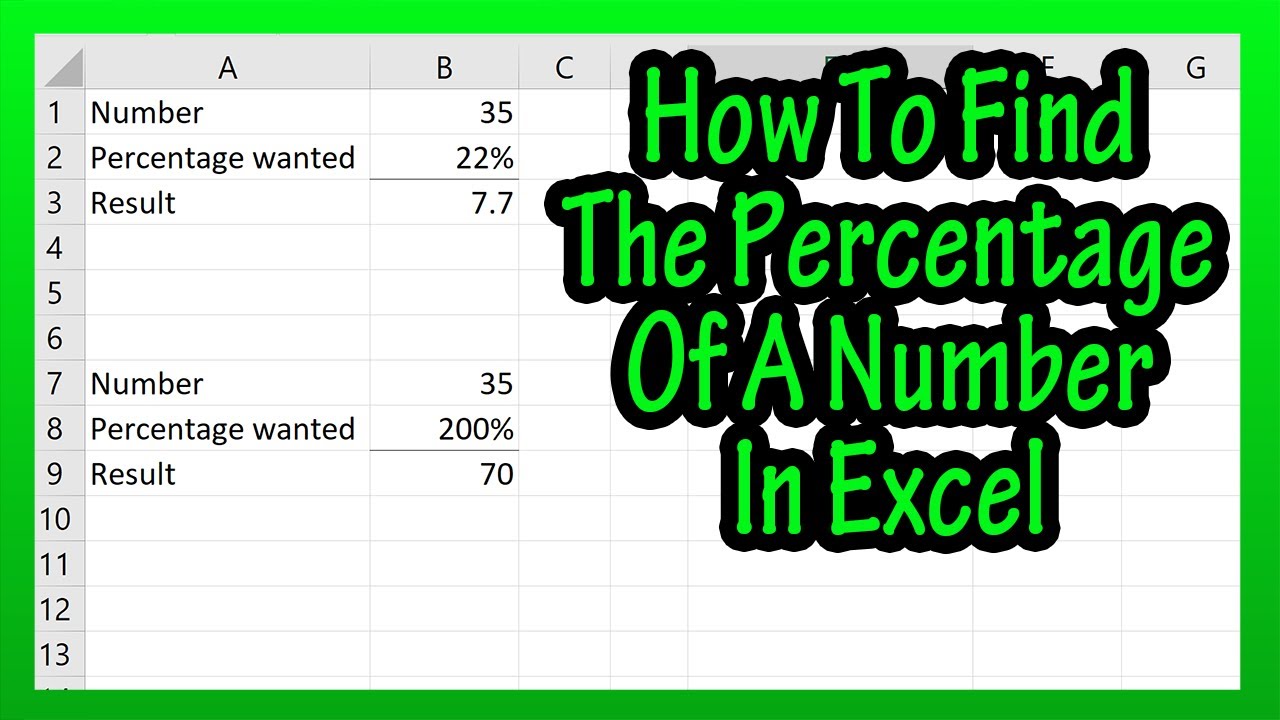
How to Find Percentage of a Number / Finding Percentage of a number / Percentage calculation - YouTube


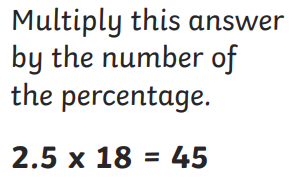

:max_bytes(150000):strip_icc()/median_definition_719-b3be9bdb33244c7fae4f582daa3dbe63.jpg)