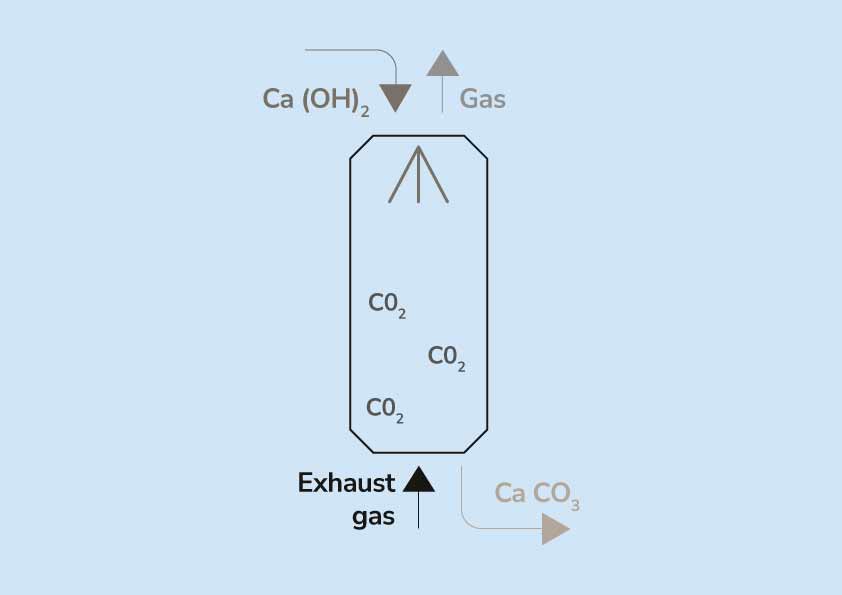
Write the balanced chemical equations for the following reaction:Calcium hydroxide + Carbon dioxide → Calcium Carbonate + Water.

Hydroxide + carbon dioxide is equal to calcium carbonate + water balance the equation numerically - Science - - 12540547 | Meritnation.com
Write the balanced chemical equations for the following reactions: i. Calcium hydroxide + Carbon dioxide → Calcium carbonate + Water - Sarthaks eConnect | Largest Online Education Community
SOLVED:When carbon dioxide is bubbled through a clear calcium hydroxide solution, the solution appears milky. Write an equation for the reaction and explain how this reaction illustrates that CO2 is an acidic

Carbon dioxide test. Test tube of limewater with carbon dioxide gas being bubbled through. The limewater, a solution of calcium hydroxide in water, is Stock Photo - Alamy

Write the balanced chemical equations for the following reactions.A Calcium hydroxide + Carbon dioxide → Calcium carbonate + waterB Zinc + Silver nitrate → Zinc nitrate + SilverC Aluminium + copper chloride

Look at Figure 4.1 and answer the following questions (a) What change would you observe in the calcium hydroxide solution taken - Question 53, 4. Carbon and its Compounds, Ncert Exemplar -

Energy-efficient chemical regeneration of AMP using calcium hydroxide for operating carbon dioxide capture process - ScienceDirect
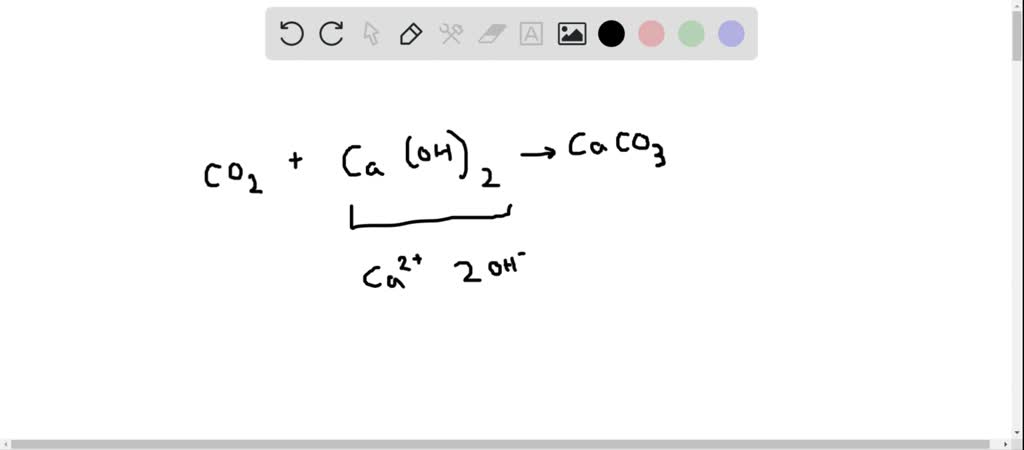
30. Ca(OH)_2 + CO_2 CaCO_3 + H_2O Why calcium hydroxide + Carbon dioxide form Calcium carbonate + Water