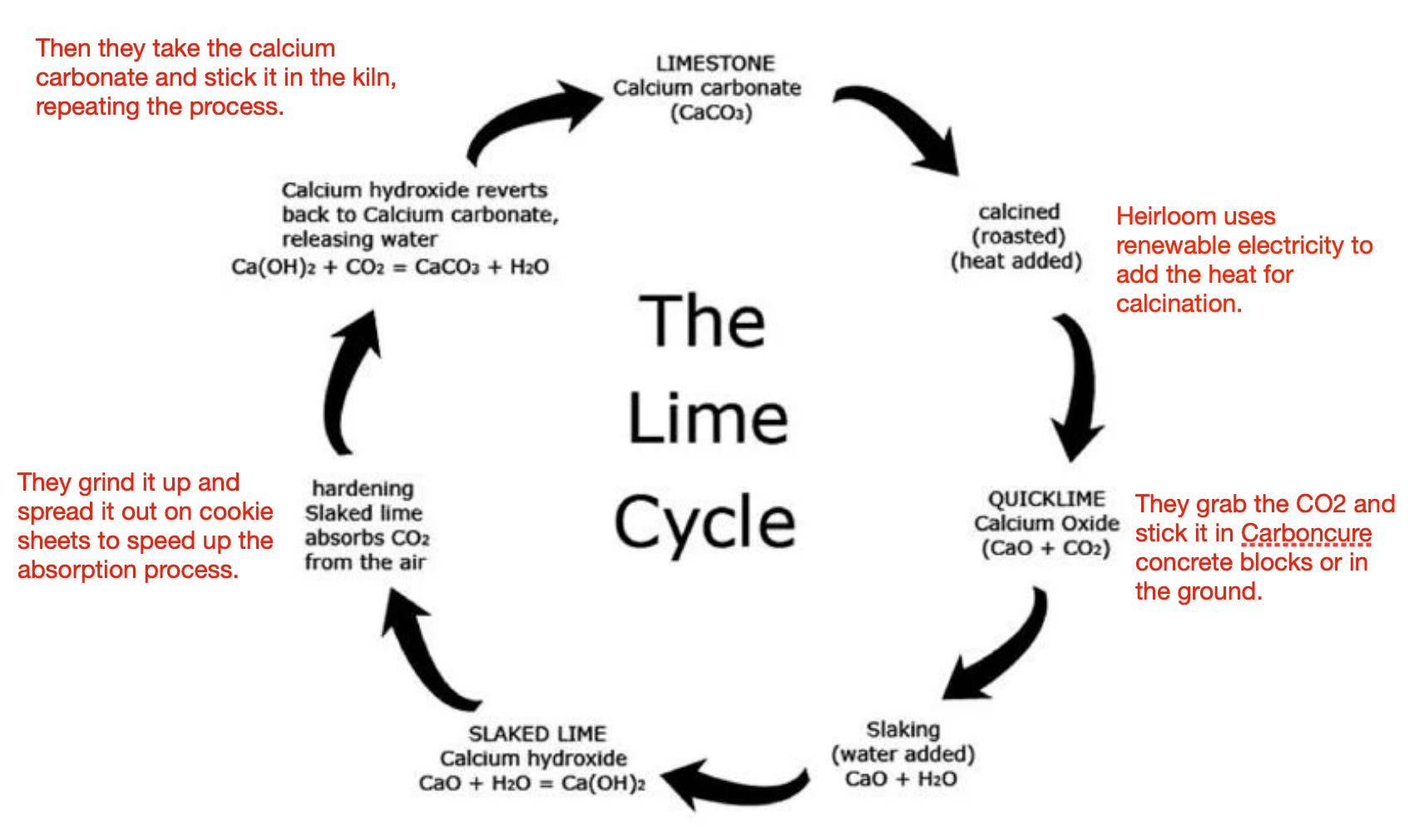
Heating Limestone (CaCO3) produces calcium oxide and carbon dioxide. The equation for this is: CaCO3(s) —> CaO(s) + CO2(g) If we heated up 300g of calcium carbonate. What mass of calcium oxide

One method of determining the proportion of calcium carbonate in a coral is to dissolve a known mass of the coral in excess acid and measure the volume of carbon dioxide formed.

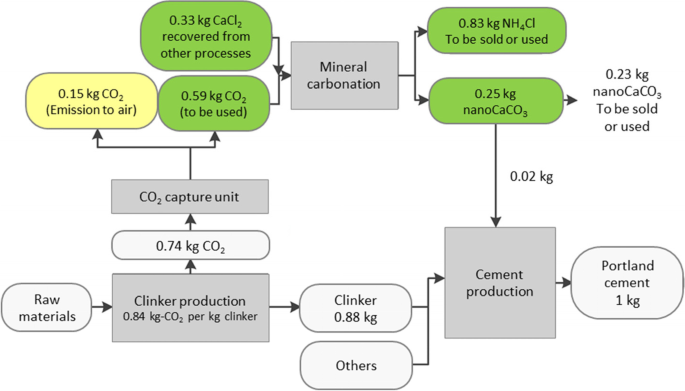
Case study for production of calcium carbonate from carbon dioxide in flue gases and steelmaking slag - ScienceDirect

Co-utilisation of CO2 and Calcium Silicate-rich Slags for Precipitated Calcium Carbonate Production (Part II) | Semantic Scholar

Carbon dioxide conversion into calcium carbonate nanoparticles using membrane gas absorption - ScienceDirect
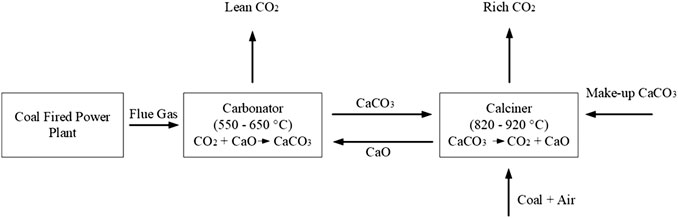
Frontiers | CO2 Capture for Dry Reforming of Natural Gas: Performance and Process Modeling of Calcium Carbonate Looping Using Acid Based CaCO3 Sorbent

Reaction steps involved in the formation of the CaCO3 sol from calcium... | Download Scientific Diagram
30. Calcium carbonate reacts with HCl to give CaCl,and CO2 according to the reaction:+ 2HCI(aq) CaCl, (aq) + CO2(g)CaCO3(s)H20(I) What mass of 20

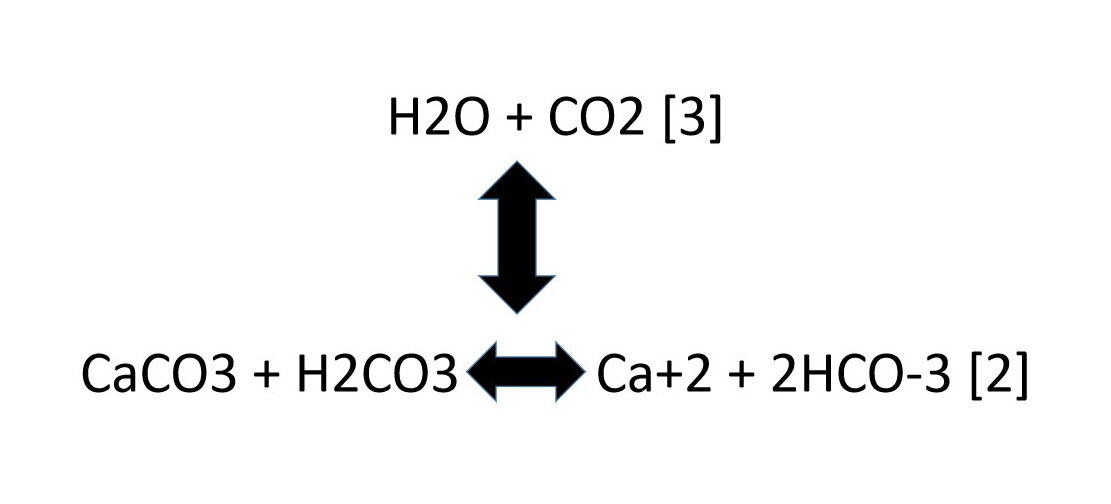
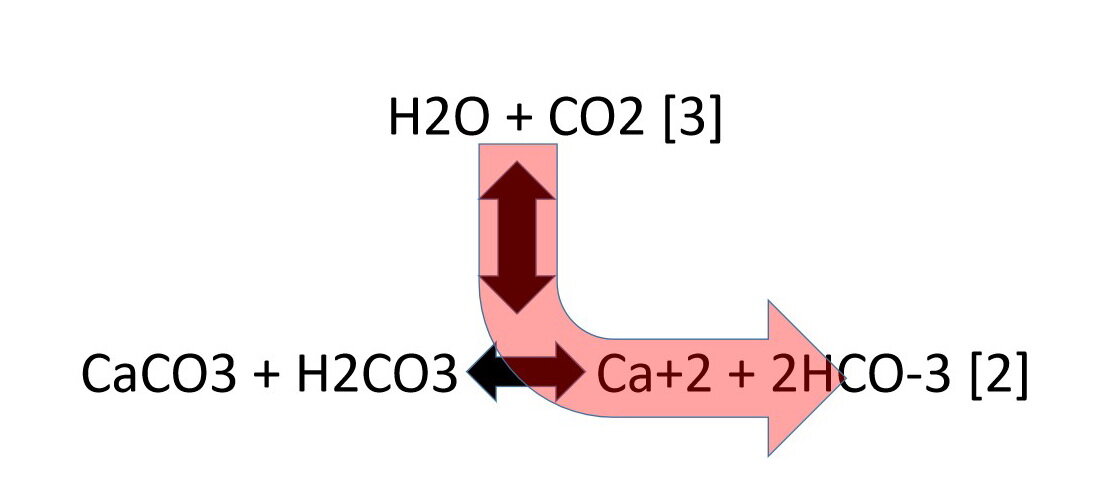
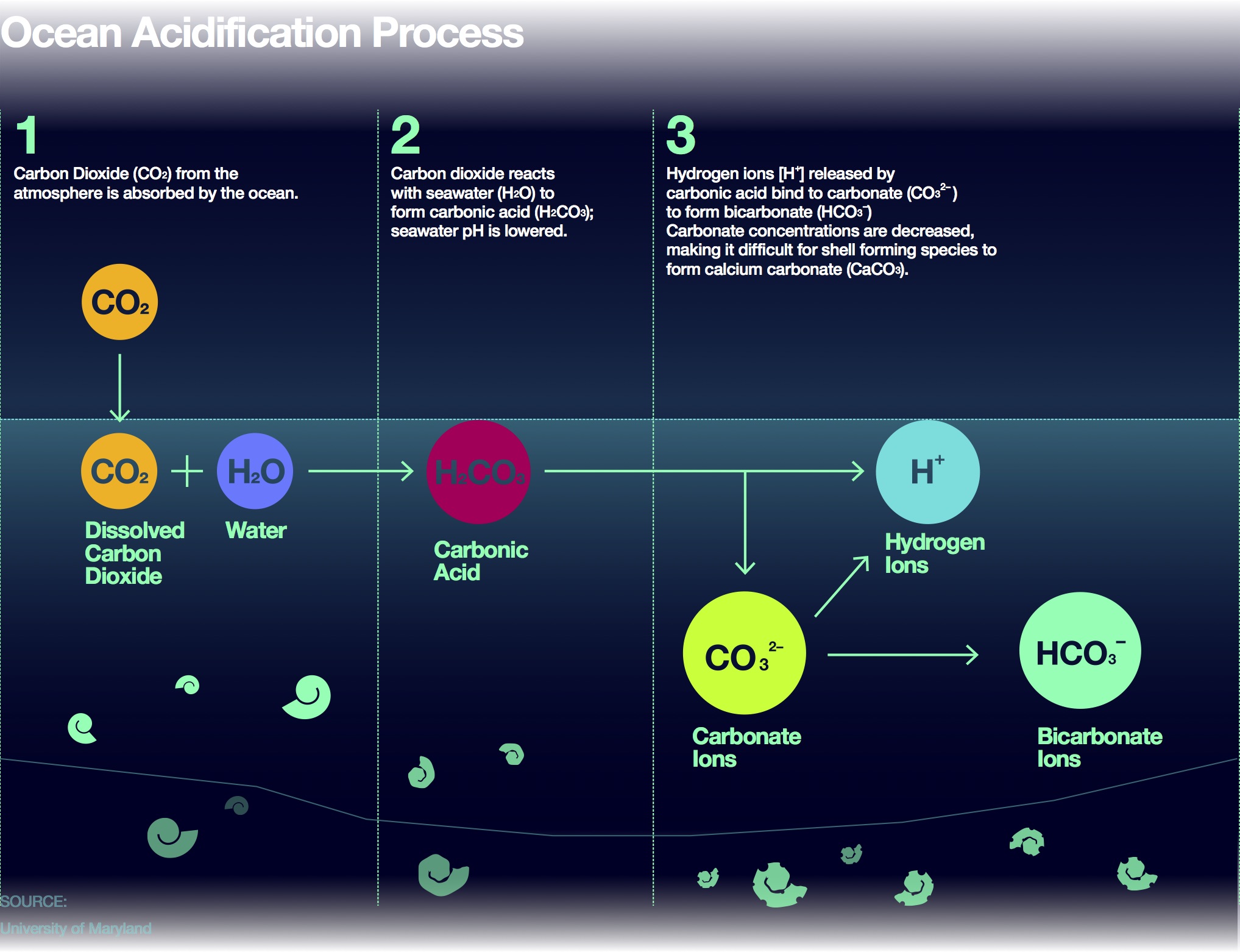
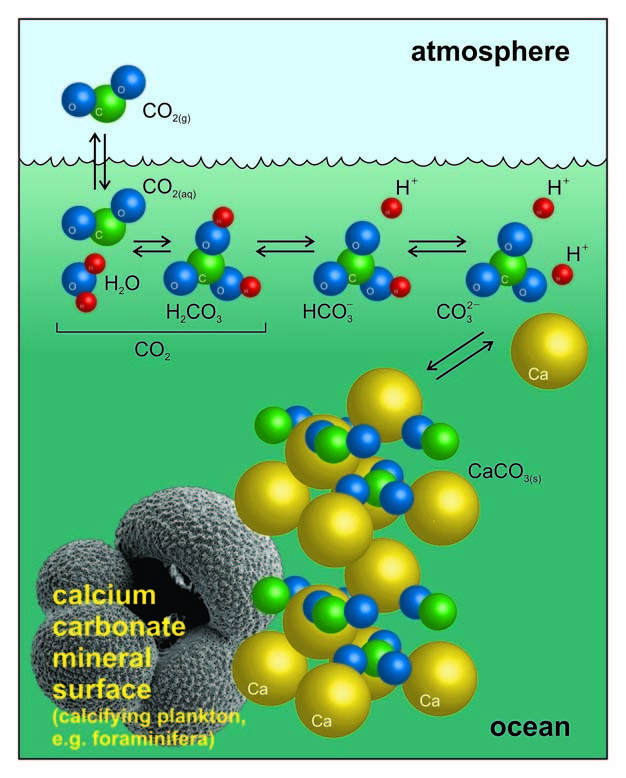
Summary of the reactions between carbon dioxide (CO2) with water (H2O)... | Download Scientific Diagram