AnanoDot, material used is Aluminum oxide doped with Carbon (Al 2 O 3... | Download Scientific Diagram
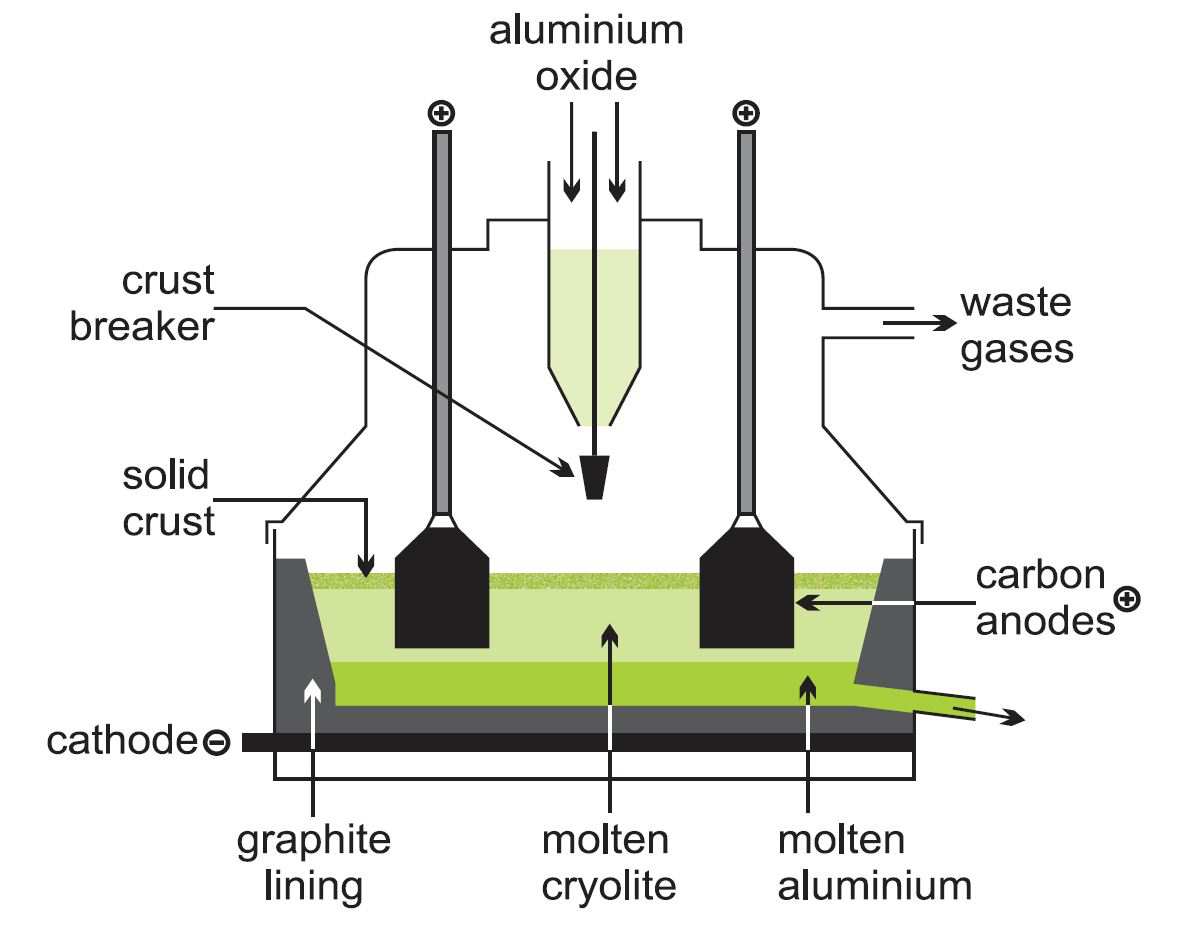
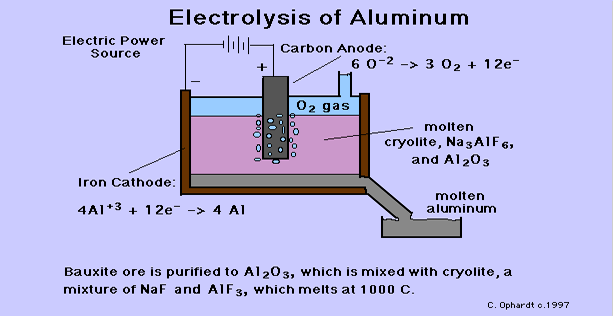
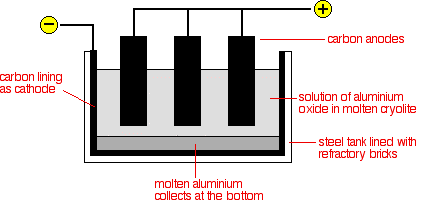
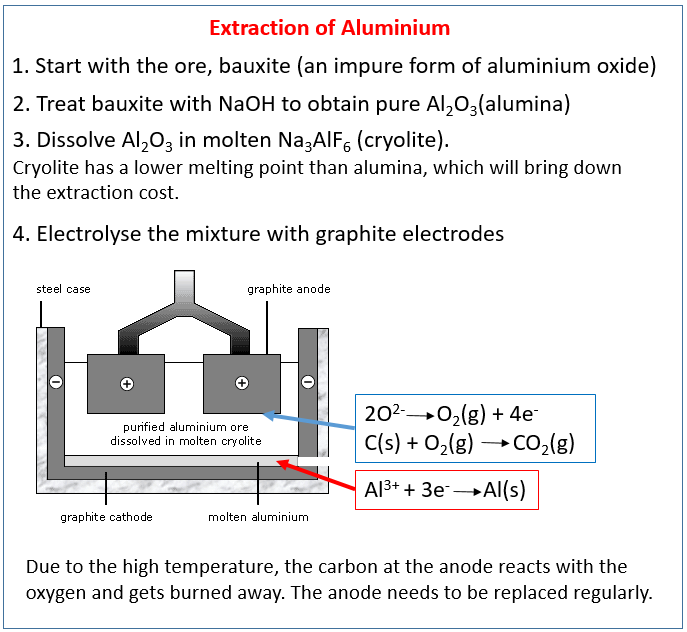
GCSE CHEMISTRY - Extraction of Aluminium - Electrolysis - Ionic Equations - Anode Replacement - GCSE SCIENCE.

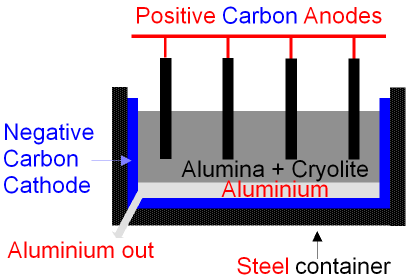
5.2 describe and explain the extraction of aluminium from purified aluminium oxide by electrolysis – igcse2016

Aluminium oxide contains 52 9% aluminium and carbon dioxide contain 27 27% carbon assuming the law of reciprocal proportional calculate - Chemistry - Some Basic Concepts of Chemistry - 12906447 | Meritnation.com
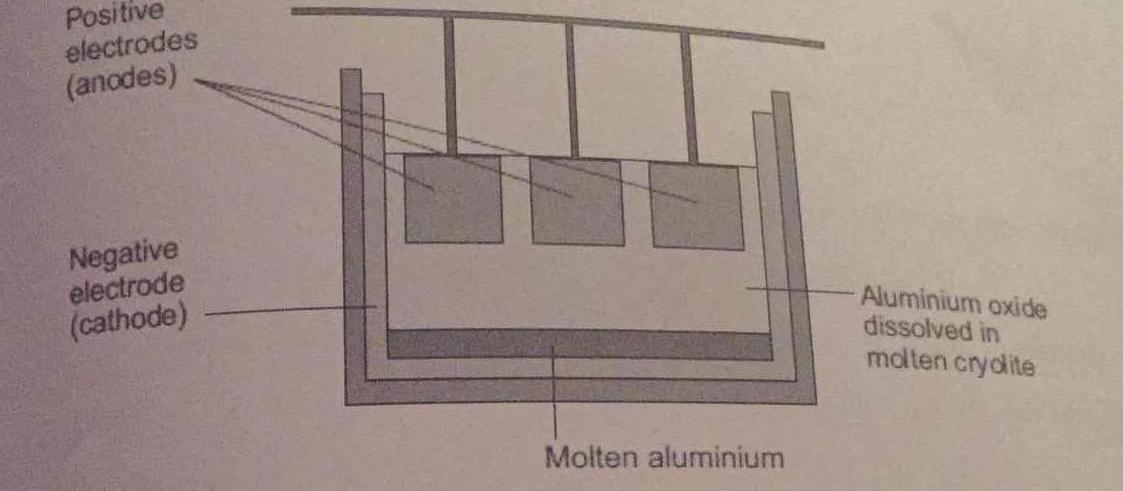
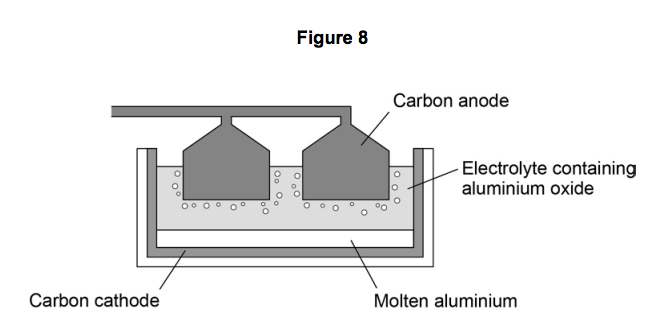
Aluminium is extracted by electrolysis, as shown in the figure. Why can aluminium not be extracted by heating aluminium oxide with carbon? | Socratic

Chemistry world - Extracting aluminium from bauxite Introduction Aluminium is too high in the electrochemical series (reactivity series) to extract it from its ore using carbon reduction. The temperatures needed are too