Therapeutic monitoring of adalimumab at non-trough levels in patients with inflammatory bowel disease | PLOS ONE
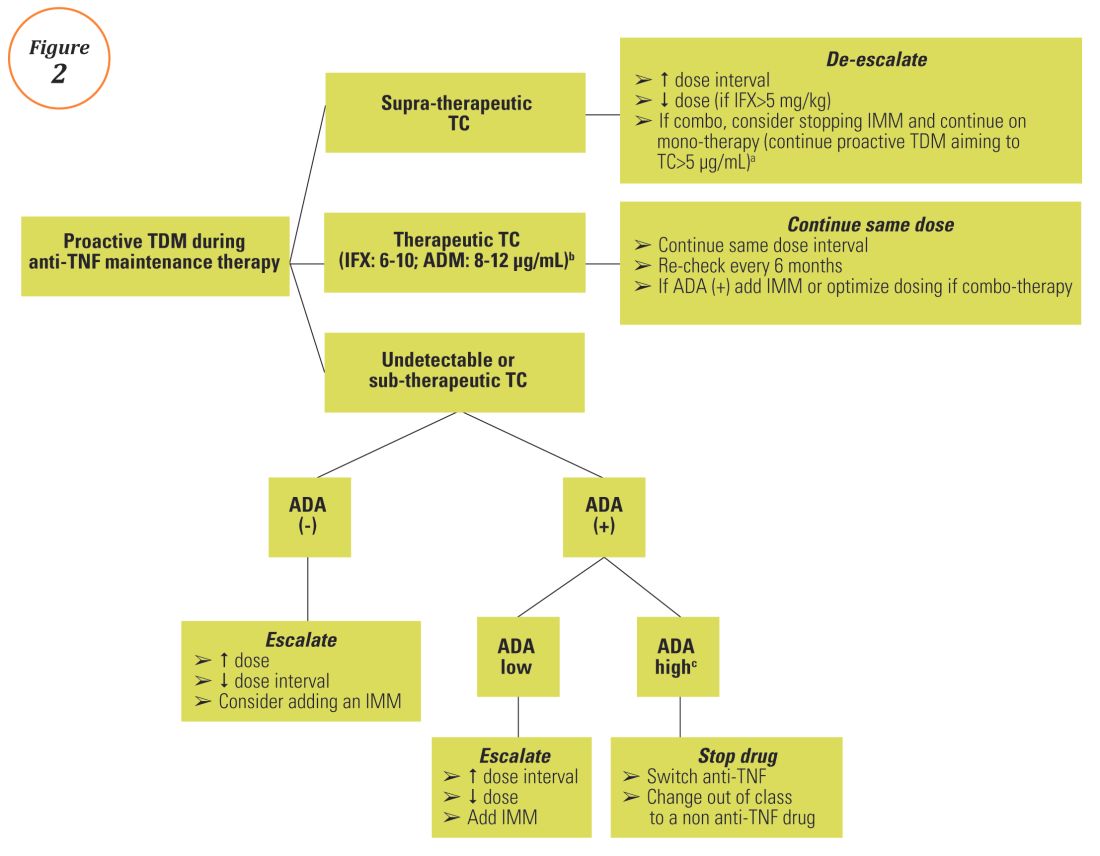
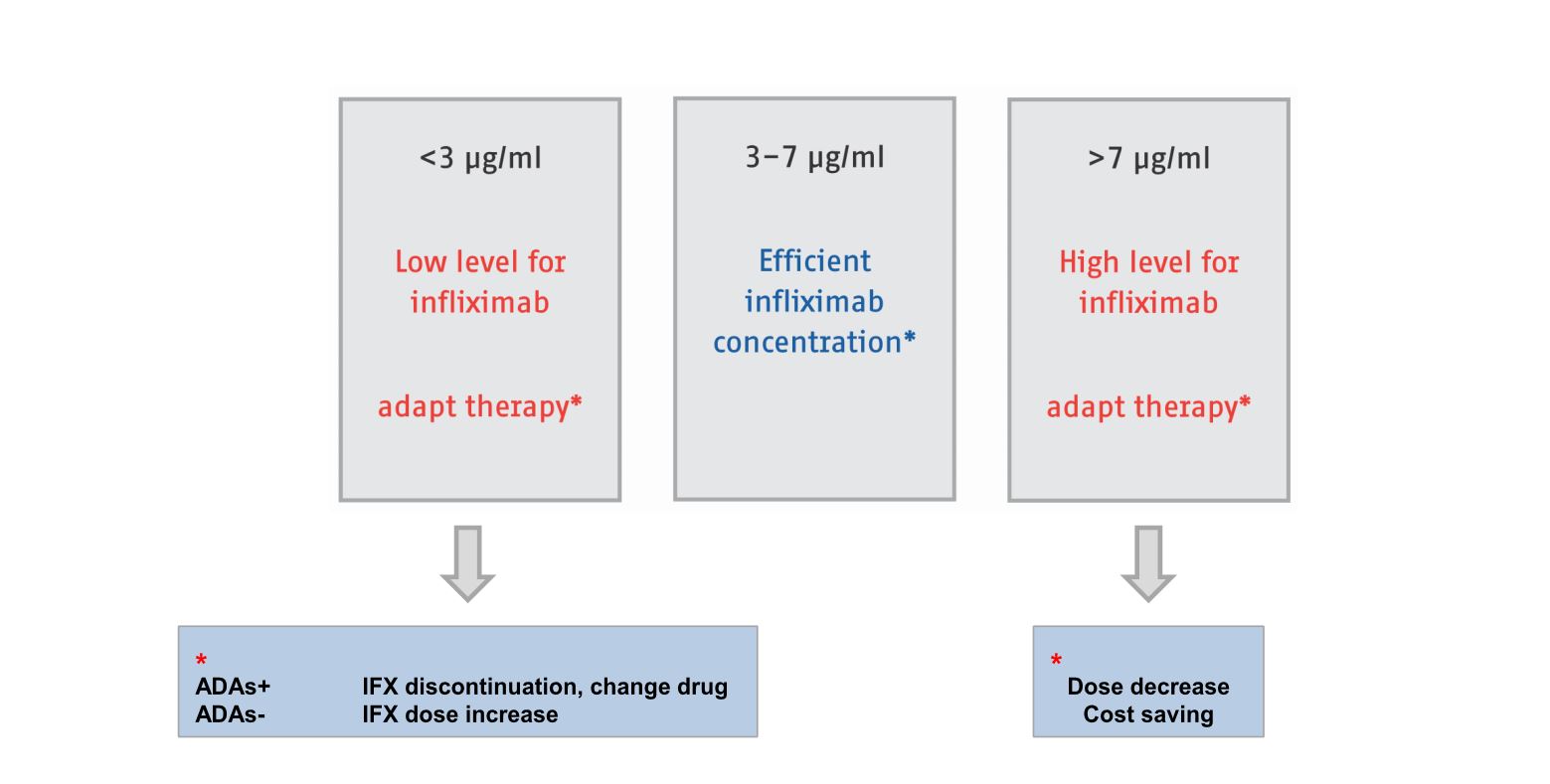
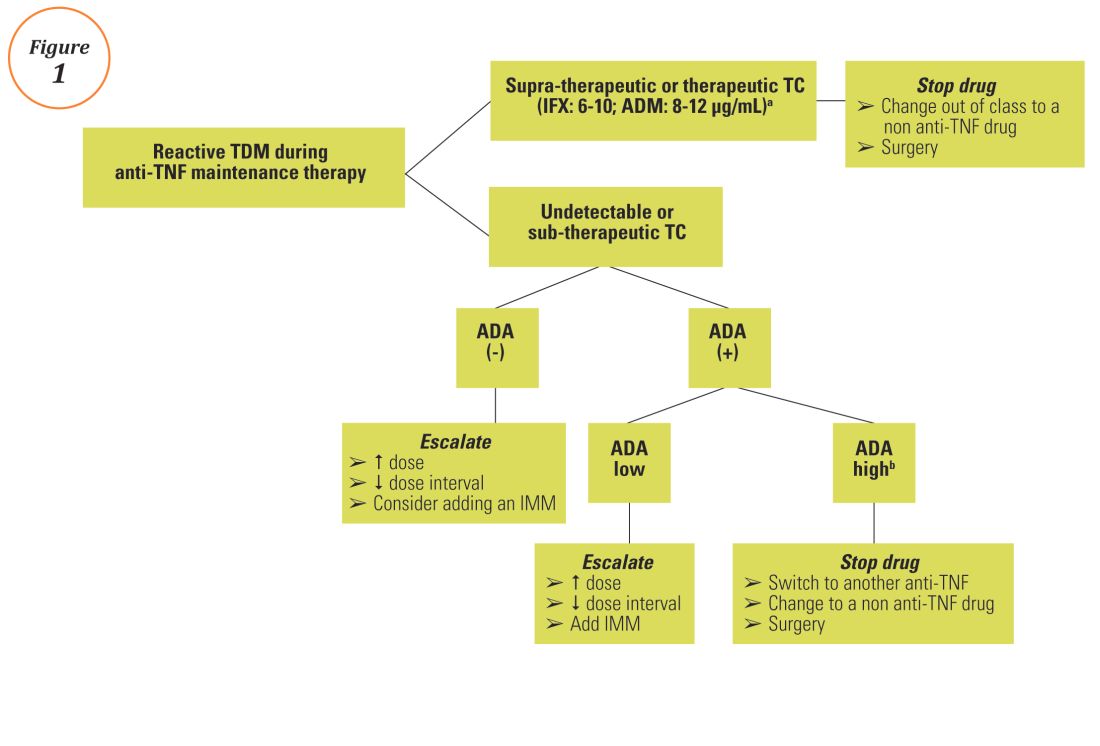
1 Scottish Biologic Therapeutic Drug Monitoring Service –Gastroenterology Guidance The purpose of this document is to provide
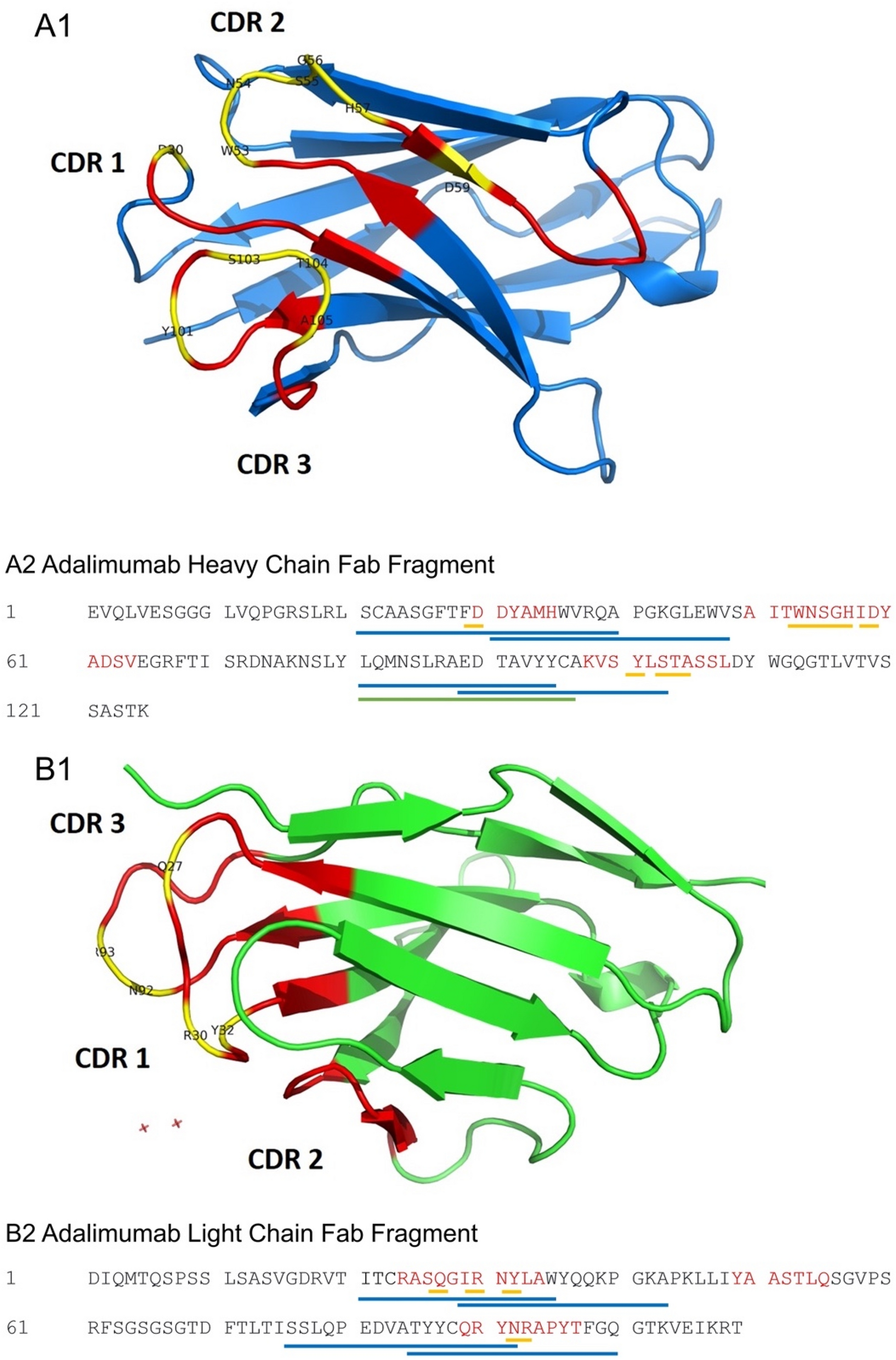
A peptide-based anti-Adalimumab antibody assay to monitor immune response to biologics treatment in juvenile idiopathic arthritis and childhood chronic non-infectious uveitis | Scientific Reports
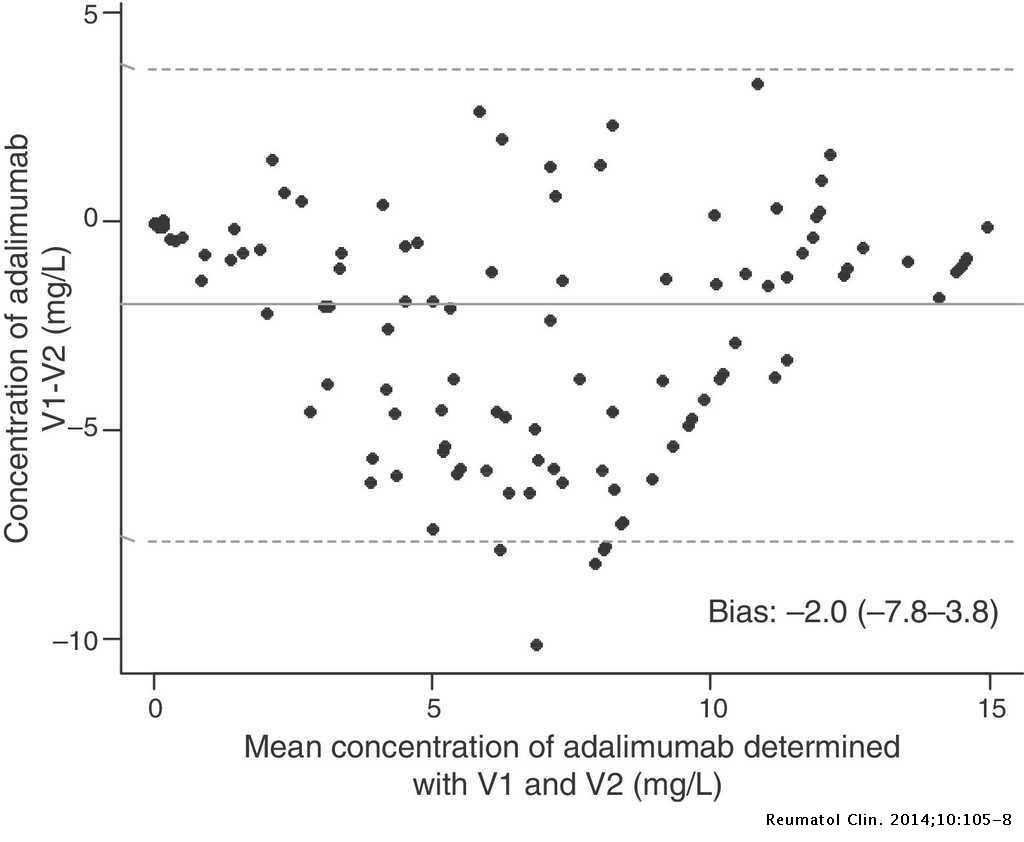
Comparative Study of Both Versions of an Immunoassay Commercialized for Therapeutic Drug Monitoring of Adalimumab in Rheumatoid Arthritis | Reumatología Clínica
Therapeutic monitoring of adalimumab at non-trough levels in patients with inflammatory bowel disease | PLOS ONE

Frontiers | Clinical Evaluation of Humira® Biosimilar ONS-3010 in Healthy Volunteers: Focus on Pharmacokinetics and Pharmacodynamics

Dynamics of circulating TNF during adalimumab treatment using a drug-tolerant TNF assay | Science Translational Medicine

Comparison between adalimumab introduction and methotrexate dose escalation in patients with inadequately controlled psoriatic arthritis (CONTROL): a randomised, open-label, two-part, phase 4 study - The Lancet Rheumatology
Frontiers | The First WHO International Standard for Adalimumab: Dual Role in Bioactivity and Therapeutic Drug Monitoring

Monitoring of adalimumab and antibodies-to-adalimumab levels in patient serum by the homogeneous mobility shift assay - ScienceDirect
Immunopharmacologic Monitoring of Therapeutic Serum Antibodies I. Policy Description II. Related Policies III. Indications and/o